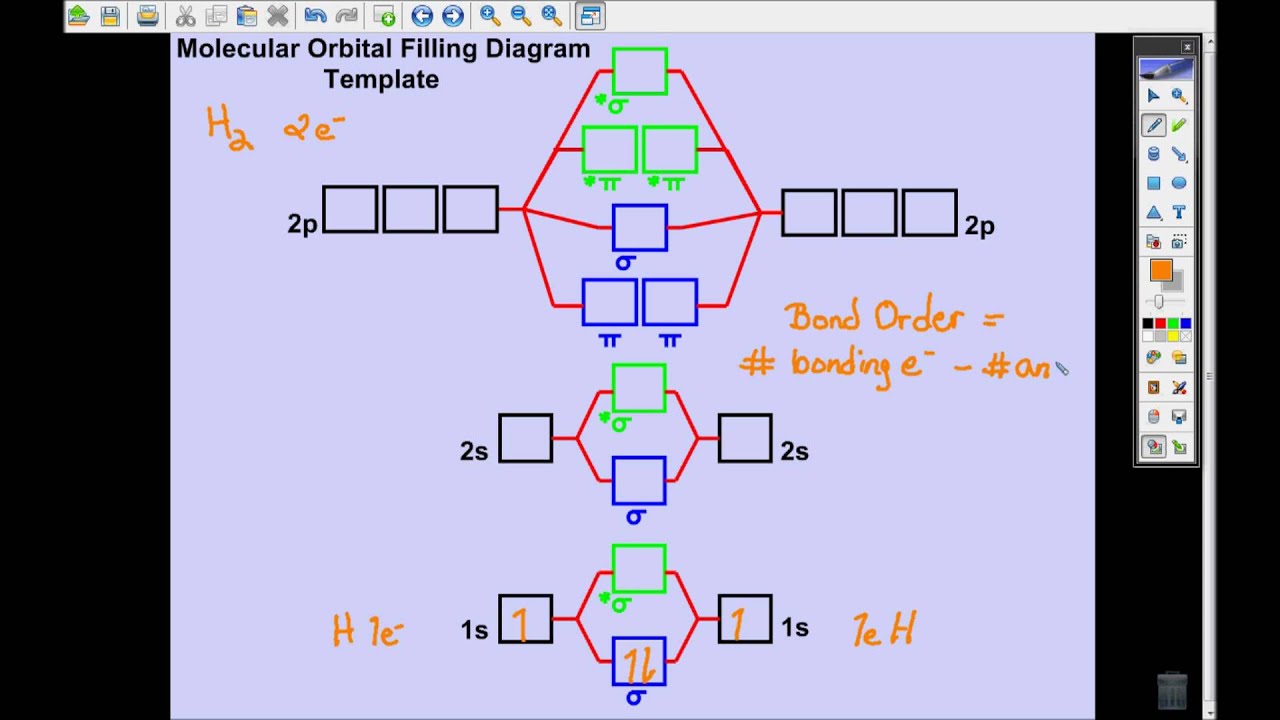
Khan Academy Molecular Orbital Theory
Khan Academy Molecular Orbital Theory - In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. When a nitrogen atom forms an ammonia. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. This is a great question that goes into the details of molecular orbital theory and chemical bonding. If you're seeing this message, it means we're having trouble loading external resources on our website. In mo theory explaining bonding, anti bonding, and non bonding orbitals in general and how to fill the electrons in the orbitals. If you're behind a web filter, please.
When a nitrogen atom forms an ammonia. In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. In mo theory explaining bonding, anti bonding, and non bonding orbitals in general and how to fill the electrons in the orbitals. This is a great question that goes into the details of molecular orbital theory and chemical bonding. If you're seeing this message, it means we're having trouble loading external resources on our website. If you're behind a web filter, please.
Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. If you're behind a web filter, please. If you're seeing this message, it means we're having trouble loading external resources on our website. In mo theory explaining bonding, anti bonding, and non bonding orbitals in general and how to fill the electrons in the orbitals. This is a great question that goes into the details of molecular orbital theory and chemical bonding. When a nitrogen atom forms an ammonia.
The Complete Guide to Understanding Molecular Orbital Diagrams on Khan
This is a great question that goes into the details of molecular orbital theory and chemical bonding. If you're behind a web filter, please. If you're seeing this message, it means we're having trouble loading external resources on our website. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. In molecular orbital theory, a covalent.
Molecular Orbital Diagram Khan Academy Hanenhuusholli
Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. In mo theory explaining bonding, anti bonding, and non bonding orbitals in general and how to fill the electrons in the orbitals. This is a great question that goes into the details of molecular orbital theory and chemical bonding. When a nitrogen atom forms an ammonia..
Understanding Molecular Orbital Diagrams with Khan Academy
If you're behind a web filter, please. If you're seeing this message, it means we're having trouble loading external resources on our website. In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy..
how to draw molecular orbital diagram khan academy Lourie Thatcher
When a nitrogen atom forms an ammonia. This is a great question that goes into the details of molecular orbital theory and chemical bonding. In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan.
Molecular Orbital Theory (MOT)→ molecular orbital theory explains abou..
In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. This is a great question that goes into the details of molecular orbital theory and chemical bonding. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. If you're behind a web filter,.
Understanding Khan Academy's Orbital Diagrams A Comprehensive Guide
If you're seeing this message, it means we're having trouble loading external resources on our website. In mo theory explaining bonding, anti bonding, and non bonding orbitals in general and how to fill the electrons in the orbitals. This is a great question that goes into the details of molecular orbital theory and chemical bonding. In molecular orbital theory, a.
Molecular Orbital Theory, Part 2 YouTube
This is a great question that goes into the details of molecular orbital theory and chemical bonding. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. If you're seeing this message, it means we're having trouble loading external resources on our website. If you're behind a web filter, please. In molecular orbital theory, a covalent.
Molecular Orbital Diagram Khan Academy Hanenhuusholli
In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. If you're seeing this message, it means we're having trouble loading external resources on our website. If you're behind a web filter, please..
3 Pi Bonds And Sp2 Hybridized Orbitals Video Khan Academy Khan Academy
In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. This is a great question that goes into the details of molecular orbital theory and chemical bonding. When a nitrogen atom forms an ammonia. In mo theory explaining bonding, anti bonding, and non bonding orbitals in general.
The Ultimate Guide to Understanding Orbital Diagrams A Khan Academy
When a nitrogen atom forms an ammonia. In mo theory explaining bonding, anti bonding, and non bonding orbitals in general and how to fill the electrons in the orbitals. Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. If you're behind a web filter, please. If you're seeing this message, it means we're having trouble.
In Mo Theory Explaining Bonding, Anti Bonding, And Non Bonding Orbitals In General And How To Fill The Electrons In The Orbitals.
Explore advanced applications of molecular orbital theory in this comprehensive guide on khan academy. In molecular orbital theory, a covalent bond is formed whenever two atoms overlap all of their orbitals, regardless of whether they are valence. When a nitrogen atom forms an ammonia. If you're behind a web filter, please.
This Is A Great Question That Goes Into The Details Of Molecular Orbital Theory And Chemical Bonding.
If you're seeing this message, it means we're having trouble loading external resources on our website.